Life-Saving Medication For The Most SEVERE Depression Exists — So Where Is It?!
As one doctor says, "How do you not offer this drug to people?"
 pexels
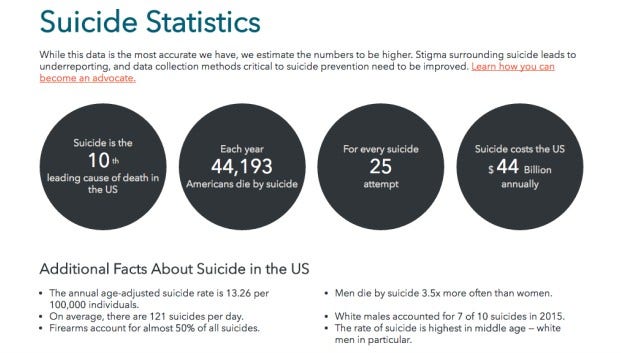
pexels Depression causes a myriad of symptoms, both physical and mental for the more than 15 million Americans (that's almost 7% of the population) who suffer from it.
And according to WebMD, over 90% of people who die by suicide have clinical depression.
Just look at these numbers!
I'm not going to pretend that my life is filled with lilies and roses. I myself have attempted suicide. Sometimes the pain is just too much, and you'd rather die than continue to go on. That's the truth of it. It took three years of trial and error working with medications that made my stomach burn and my head feel it was being stabbed with a knife, to find the right cocktail to stabilize me.
Why? Because sometimes the side effects of anti-depressants are even worse than the pain of a depressive episode. And yet sometimes, unexpected side effects can shock the medical establishment with unforeseen benefits.
Am I 100% better now? Hell, no! It's a lifelong illness that I've come to accept. Without those medications, I'd fail to function at life. Though I still have my bouts with suicidal ideation, they are few and far between, but what about those who are only a step away from self-harm or suicide?
As it turns out, there may be new hope on the horizon ...
Ketamine — better known on the streets as "Special K" — has now been given to 3,000 patients suffering from severe depression. Normally prescribed as an anesthetic, doctors are beginning to prescribe it "off-label," which means using it for the benefits its side effects and not its intended use.
Other doctors are worried there haven't been enough studies done to see its long term effect on those who take it regularly.
Professor of Psychiatry at Yale University, Gerard Sanacora, who uses Ketamine to treat patients, recently told NPR that he is often asked how he could prescribe it without knowing more about the drug. He responds:
"If you have patients that are likely to seriously injure themselves or kill themselves within a short period of time, and they've tried the standard treatments, how do you not offer this treatment?"
An assistant professor of psychiatry and neuroscience at the Icahn School of Medicine at Mt. Sinai in New York, James Murrough, also told NPR:
"We haven't had large-scale trials. We don't know how much or how often it should be given for it to be effective or safe … There’s warranted caution that's balanced with an optimism that says we've never had a new medication for depression since the era of Prozac."
According to the Mayo Clinic, Prozac (generic name Fluoxetine) is an antidepressant that belongs to a group of medicines known as selective serotonin reuptake inhibitors (SSRIs). These medications work by increasing the activity of a chemical called serotonin in the brain. Unlike Prozac and other SSRIs, Ketamine works on a different neurotransmitter: glutamate.
What's interesting is that the FDA is currently trying to approve a breakthrough therapy designation on a chemical relative to Ketamine, called Esketamine. Sanacora says, "This is probably the most interesting and exciting new development that I've seen in my career, and probably going back over the past 50 to 60 years.”
According to a press release from Johnson & Johnson:
"Janssen Research & Development, LLC, one of the Janssen Pharmaceutical Companies of Johnson & Johnson, announced today that the U.S. Food and Drug Administration (FDA) has granted a Breakthrough Therapy Designation for esketamine, an investigational antidepressant medication, for the indication of major depressive disorder with imminent risk for suicide. If approved by the FDA, esketamine would be one of the first new approaches to treat major depressive disorder available to patients in the last 50 years.
This also marks the second time esketamine has received a Breakthrough Therapy Designation from the U.S. regulatory authority. Esketamine was first granted this designation for treatment-resistant depression in November 2013. Breakthrough Therapy Designation is intended to expedite development and review timelines when preliminary clinical evidence indicates the drug may demonstrate substantial improvement on one or more clinically significant endpoints over available therapies for serious or life-threatening conditions."
In essence, all these fancy words mean we may soon have access to a life-saving drug that could finally conquer depression so severe it can result in self-harm, or worse.

PopKey
I've been down the road that leads to the darkness of depression. I've been down the road that ends in the fork of the choice between life or death. All I can say with certainty is that I wish I had the option to ease the excruciating pain that leads you to that place.
I'm thrilled to know doctors are trying to improve the medication available to the mental health community. This not only validates our illness (which is constantly dismissed and stigmatized by those who don't understand it) and potentially opens up a new door to hope and peace for those who suffer.
I always hear that it gets better. I never believed it, but now I'm starting to see the glimmer of hope.
We are not alone.
To learn more about the potential of Ketamine to help those who suffer from depression and PTSD, watch this TEDx Talk by neuroscientist, writer and entrepreneur Rebecca Brachman.